A) When two p orbitals of similar phase overlap side-by-side,a p* antibonding molecular orbital is formed.
B) When two p orbitals of opposite phase overlap side-by-side,a p bonding molecular orbital is formed.
C) A p bonding molecular orbital is higher in energy than the two atomic p orbitals from which it is formed.
D) A p* antibonding molecular orbital is higher in energy than the two atomic p orbitals from which it is formed.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 13CNMR signals does the following compound exhibit? 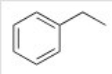
A) 4
B) 5
C) 6
D) 8
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What orbitals are used to form the indicated bond? 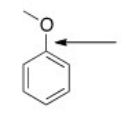
A) sp3
B) sp2
C) p
D) sp3 and sp2
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the following compound not aromatic? 
A) It has 4n electrons.
B) It is not cyclic.
C) It has 4n+2 electrons.
D) The pi electron system is not continuous.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 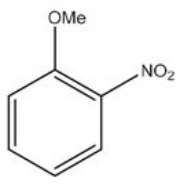
A) o-nitroanisole
B) m-nitroanisole
C) p-nitroanisole
D) 2-nitroaniline
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following heterocycles is not aromatic? 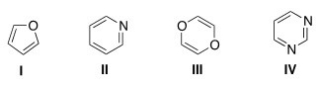
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 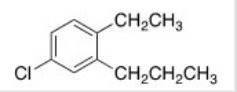
A) 1-Chloro-4-ethyl-3-propylbenzene
B) 1-Chloro-4-ethyl-5-propylbenzene
C) 4-Chloro-1-ethyl-2-propylbenzene
D) 4-Chloro-2-propyltoluene
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 47 of 47
Related Exams