A) none
B) one
C) two
D) three
E) four
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For potassium metal, the work function 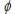 (the minimum energy needed to eject an electron from the metal surface) is 3.68 × 10-19 J. Which is the longest wavelength of the following which could excite photoelectrons?
(the minimum energy needed to eject an electron from the metal surface) is 3.68 × 10-19 J. Which is the longest wavelength of the following which could excite photoelectrons?
A) 550. nm
B) 500. nm
C) 450. nm
D) 400. nm
E) 350. nm
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In the quantum mechanical treatment of the hydrogen atom, the energy depends on the principal quantum number n but not on the values of l or ml.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the photoelectric effect, a photon with an energy of 5.3 × 10-19 J strikes an electron in a metal. Of this energy, 3.6 × 10-19 J is the minimum energy required for the electron to escape from the metal. The remaining energy appears as kinetic energy of the photoelectron. What is the velocity of the photoelectron, assuming it was initially at rest?
A) 3.7 × 1014 m/s
B) 3.7 × 1011 m/s
C) 1.9 × 106 m/s
D) 6.1 × 105 m/s
E) 1.7 × 10-19 m/s
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The Rydberg equation, giving the wavelengths of lines in the spectrum of the hydrogen atom, was obtained by assuming that energy is quantized.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Who proposed a model that successfully explained the photoelectric effect?
A) Planck
B) Einstein
C) Compton
D) Rydberg
E) Bohr
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the quantum mechanical treatment of the hydrogen atom, which one of the following combinations of quantum numbers is not allowed? 
A) A
B) B
C) C
D) D
E) E
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
In the quantum mechanical treatment of the hydrogen atom, the probability of finding an electron at any point is proportional to the wave function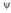 .
.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the arrangement of electromagnetic radiation which starts with the lowest energy and increases to greatest energy.
A) radio, visible, infrared, ultraviolet
B) infrared, visible, ultraviolet, microwave
C) visible, ultraviolet, infrared, gamma rays
D) X-radiation, visible, infrared, microwave
E) microwave, infrared, visible, ultraviolet
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Excited hydrogen atoms radiate energy in the
A) infrared region only.
B) visible region only.
C) ultraviolet region only.
D) visible and ultraviolet regions only.
E) infrared, visible, and ultraviolet regions.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the Rydberg equation, the line with the shortest wavelength in the emission spectrum of atomic hydrogen is predicted to lie at a wavelength (in nm) of
A) 91.2 nm.
B) 1.10 × 10-2 nm.
C) 1.10 × 102 nm.
D) 1.10 × 1016 nm.
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Other factors being constant, a heavy object will have a longer de Broglie wavelength than a light object.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which scientist first proposed that particles of matter could have wave properties?
A) Einstein
B) Planck
C) de Broglie
D) Compton
E) Heisenberg
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Electromagnetic radiation of 500 nm wavelength lies in the __________ region of the spectrum.
A) infrared
B) visible
C) ultraviolet
D) X-ray
E) ![]() -ray
-ray
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Who was the first scientist to propose that an object could emit only certain amounts of energy?
A) Planck
B) Einstein
C) Bohr
D) Rydberg
E) de Broglie
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the arrangement of electromagnetic radiation which starts with the lowest energy and increases to greatest energy.
A) radio, infrared, ultraviolet, gamma rays
B) radio, ultraviolet, infrared, gamma rays
C) gamma rays, infrared, radio, ultraviolet
D) gamma rays, ultraviolet, infrared, radio
E) infrared, ultraviolet, radio, gamma rays
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The size of an atomic orbital is associated with
A) the principal quantum number (n) .
B) the angular momentum quantum number (l) .
C) the magnetic quantum number (ml) .
D) the spin quantum number (ms) .
E) the angular momentum and magnetic quantum numbers, together.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which word best describes the phenomenon which gives rise to a rainbow?
A) reflection
B) dispersion
C) diffraction
D) interference
E) deflection
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a correct set of quantum numbers for an electron in a 5f orbital?
A) n = 5, l = 3, ml = +1
B) n = 5, l = 2, ml = +3
C) n = 4, l = 3, ml = 0
D) n = 4, l = 2, ml = +1
E) n = 5, l = 4, ml = 3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A radio wave has a frequency of 8.6 × 108 Hz. What is the energy of one photon of this radiation?
A) 7.7 × 10-43 J
B) 2.3 × 10-34 J
C) 5.7 × 10-25 J
D) 1.7 × 10-16 J
E) > 10-15 J
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 69
Related Exams