A) dipole-dipole forces
B) London dispersion forces
C) ion-dipole forces
D) covalent bonding
E) ion-ion forces
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Solids are generally most stable in crystalline form.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane is a component of fuel used in internal combustion engines. The dominant intermolecular forces in octane are
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) covalent bonds.
E) carbon-hydrogen bonds.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pure substances would be expected to have the highest boiling point?
A) CH4
B) Cl2
C) Kr
D) CH3Cl
E) N2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of the unit cell for nickel(II) oxide. How many O2- anions are there in the unit cell? 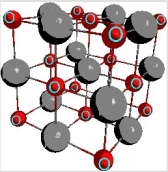
A) 1
B) 4
C) 8
D) 13
E) 14
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following pure substances will exhibit hydrogen bonding?
A) CH4
B) H2C=CH2
C) H2O
D) CHCl3
E) CHF3
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Which liquid is expected to have the larger surface tension at a given temperature, CCl4 or H2O? Briefly explain.
Correct Answer

verified
Water would have the...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which substance has the lowest vapor pressure at room temperature?
A) HF
B) HCl
C) HBr
D) HI
E) H2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Consider the phase diagram shown below. 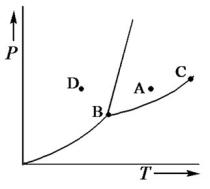 a. What phase(s) is/are present at point A?
b. What phase(s) is/are present at point B?
c. Name point C and explain its significance.
d. Starting at D, if the pressure is lowered while the temperature remains constant, describe what will happen.
a. What phase(s) is/are present at point A?
b. What phase(s) is/are present at point B?
c. Name point C and explain its significance.
d. Starting at D, if the pressure is lowered while the temperature remains constant, describe what will happen.
Correct Answer

verified
a. liquid
b. solid, liquid, and gas
c. C...View Answer
Show Answer
Correct Answer
verified
b. solid, liquid, and gas
c. C...
View Answer
True/False
Only molecules which do not have dipole moments can experience dispersion forces.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Liquid ammonia can be used as a refrigerant and heat transfer fluid. How much energy is required to heat 25.0 g of NH3(l) from -65.0°C to -12.0°C? 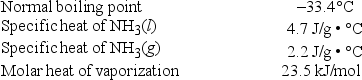
A) 4.92 kJ
B) 16.0 kJ
C) 34.5 kJ
D) 39.4 kJ
E) 182.8 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What types of intermolecular forces exist between hydrogen fluoride molecules? 
A) II and III
B) I, II, and III
C) I only
D) III only
E) I and III
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The strongest intermolecular interactions between pentane (C5H12) molecules arise from
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) ion-dipole interactions.
E) carbon-carbon bonds.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The strongest intermolecular interactions between ethyl alcohol (CH3CH2OH) molecules arise from
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) ion-dipole interactions.
E) carbon-oxygen bonds.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lead crystallizes in the face-centered cubic lattice. What is the coordination number for Pb?
A) 4
B) 6
C) 8
D) 10
E) 12
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following in order of increasing boiling point: RbCl, CH3Cl, CH3OH, CH4.
A) CH3OH < CH3Cl < RbCl < CH4
B) CH3OH < CH4 < CH3Cl < RbCl
C) RbCl < CH3 Cl < CH3 OH < CH4
D) CH4 < CH3OH < CH3Cl < RbCl
E) CH4 < CH3Cl < CH3OH < RbCl
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What name is given to the phenomenon where a thin film of water contracts and adheres to the wall of a glass cylinder?
A) Surface tension
B) Adhesion
C) Polarity
D) Capillary action
E) Cohesion
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name given to the attractive forces that hold particles together in the condensed phase?
A) ionic bonds
B) covalent bonds
C) intermolecular forces
D) electronegativity
E) electron attraction
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Ice is less dense than water due to the formation of hydrogen bonds.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which corresponds to the temperature-dependent partial pressure above the surface of a liquid?
A) Surface tension
B) Vapor pressure
C) Boiling point
D) Viscosity
E) Capillary action
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 137
Related Exams