A) the Pauli exclusion principle.
B) Bohr's equation.
C) Hund's rule.
D) de Broglie's relation.
E) Dalton's atomic theory.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
In which region of the electromagnetic spectrum would radiation of the wavelength λ = 147.3 nm be found?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the arrangement of electromagnetic radiation which starts with the shortest wavelength and increases to longest wavelength.
A) radio, infrared, ultraviolet, gamma rays
B) radio, ultraviolet, infrared, gamma rays
C) gamma rays, radio, ultraviolet, infrared
D) gamma rays, infrared, radio, ultraviolet
E) gamma rays, ultraviolet, infrared, radio
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum number of electrons in an atom that can have the following set of quantum numbers? n = 4, l = 3, ml = -2, ms = +1/2
A) 0
B) 1
C) 2
D) 6
E) 10
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
________ is the number of waves passing through a specific point per second.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many orbitals are allowed in a subshell if the angular momentum quantum number for electrons in that subshell is 3?
A) 1
B) 3
C) 5
D) 7
E) 9
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A possible set of quantum numbers for the last electron added to complete an atom of germanium in its ground state is 
A) A
B) B
C) C
D) D
E) E
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following subshells has the highest energy in the element tantalum?
A) 4s
B) 4d
C) 5p
D) 4f
E) 6s
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solar radiation spectrum peaks at a wavelength of approximately 500 nm. Calculate the energy of one photon of that radiation (c = 3.00 × 108 m/s; h = 6.63 × 10-34 J • s) .
A) 4 × 10-10 J
B) 6 × 1014 J
C) 1 × 10-27 J
D) 2 × 10-25 J
E) 4 × 10-19 J
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
For the following equations a. name the scientist to whom the equation is attributed. b. in not more than three lines, explain clearly what the equation means or represents. 1. E = nhv 2. λ = h/mu
Correct Answer

verified
1. a. Planck
b. A blackbody can only emi...View Answer
Show Answer
Correct Answer
verified
b. A blackbody can only emi...
View Answer
Multiple Choice
The de Broglie equation predicts that the wavelength (in m) of a proton moving at 1000. m/s is ________. (h = 6.63 × 10-34 J • s; mass of a proton = 1.673 × 10-24 g)
A) 3.96 × 10-10 m
B) 3.96 × 10-7 m
C) 2.52 × 106 m
D) 2.52 × 109 m
E) > 1010 m
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct electron configuration for Cu (Z = 29) .
A) [Ar]4s23d9
B) [Ar]4s13d10
C) [Ar]4s24p63d3
D) [Ar]4s24d9
E) [Ar]5s24d9
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the total number of electrons that can occupy the 4f orbitals?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many orbitals are there in the n = 4 level of the H-atom?
A) 4
B) 6
C) 8
D) 16
E) 18
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ground-state atom has an electron configuration described by the following orbital diagram? 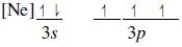
A) phosphorus
B) nitrogen
C) arsenic
D) vanadium
E) sulfur
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the wavelength, in nanometers, of the light emitted by a hydrogen atom when its electron drops from the n = 7 to the n = 4 principal energy level. Recall that the energy levels of the H atom are given by En = -2.18 × 10-18 J (1/n2) . (c = 3.00 × 108 m/s; h = 6.63 × 10-34 J • s)
A) 4.45 × 10-20 nm
B) 2.16 × 10-6 nm
C) 9.18 × 10-20 nm
D) 1.38 × 1014 nm
E) 2.17 × 103 nm
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are there in the 3rd principal energy level (n = 3) of a phosphorus atom?
A) 3
B) 5
C) 6
D) 8
E) 10
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration? [Kr]5s24d105p2
A) Sn
B) Sb
C) Pb
D) Ge
E) Te
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
________ is the quantum number which describes the shape of an atomic orbital.
Correct Answer

verified
The angula...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The electron configuration of a ground-state vanadium atom is
A) [Ar]4s24d3.
B) [Ar]4s24p3.
C) [Ar]4s23d3.
D) [Ar]3d5.
E) [Ar]4s23d7.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 135
Related Exams