A) CN-
B) I-
C) OH-
D) H2O
E) NH3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidation number of Co in the complex Co(H2O) 3Cl3
A) +1
B) +2
C) +3
D) +4
E) None of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following complexes has optical (chiral) isomers
A) [Co(H2O) 4Br2]+
B) [Co(en) Cl4]-
C) [Co(en) (H2O) 4]3+
D) [Co(en) 2Cl2]+
E) Co(H2O) 3Cl3
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the number of unpaired electrons in the [Cr(en) 3]2+ ion.
A) 0
B) 1
C) 3
D) 5
E) None of the above
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
The correct name of the complex ion [CuCl3Br(NH3)2]2- is: diamminebromotrichlorocuprate(I) ion
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound [Co(en) 2Cl2]Cl, the coordination number and oxidation number of the central atom are, respectively,
A) 4, +3.
B) 6, +2.
C) 4, +2.
D) 6, +3.
E) 4, +1.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cis-platinum complexes are
A) used in the extraction of silver and gold.
B) effective antidotes for heavy metal poisoning (e.g., Pb2+ and Hg2+) .
C) used to provide nutrients for plants.
D) used to prevent eutrophication of lakes.
E) effective antitumor agents.
G) A) and C)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
What is the oxidation number of Fe in [Fe(CN) 6]4-
A) +1
B) +2
C) +3
D) +4
E) None of the above
G) A) and E)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Assuming a coordination complex is formed with Fe2+ and 1,10-phenanthroline (shown below) , which of the following statements is true 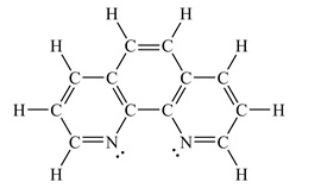
A) If two 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 2.
B) If two 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 6.
C) If three 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 3.
D) If three 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 6.
E) If four 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 4.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the number of unpaired electrons in the [Fe(CN) 6]4- ion.
A) 0
B) 1
C) 3
D) 5
E) None of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons are there in the complex ion [Mn(CN) 6]3-
A) 0
B) 1
C) 2
D) 3
E) 4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The terms that describe the geometric isomers that are possible for the complex [CrF2Cl4]3- are cis and trans.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of electrons in the 3d orbitals of a copper atom is
A) 6.
B) 7.
C) 8.
D) 9.
E) 10.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound K2[Co(en) Cl4], the coordination number (C.N.) and oxidation number (O.N.) of cobalt are
A) C.N. = 6; O.N. = +2.
B) C.N. = 6; O.N. = +3.
C) C.N. = 5; O.N. = +2.
D) C.N. = 5; O.N. = +4.
E) C.N. = 4; O.N. = +3.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Bidentate and polydentate ligands are also called chelating agents.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following complexes has optical (chiral) isomers
A) [Co(NH3) 4Br2]+
B) [Co(en) 3]3+
C) Co(NH3) 3Br3
D) [Co(en) Br4]-
E) [Co(en) (NH3) 4]3+
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the complex ion [ML6]n+, Mn+ has six d electrons and L is a weak field ligand. According to crystal field theory, the magnetic properties of the complex ion correspond to how many unpaired electrons
A) 0
B) 1
C) 2
D) 3
E) 4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of electrons in the 3d orbitals of Co3+ is
A) 4.
B) 5.
C) 6.
D) 7.
E) 10.
G) B) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
In the complex ion [ML6]n+, Mn+ has five d electrons and L is a strong field ligand. According to crystal field theory, the magnetic properties of the complex ion correspond to how many unpaired electrons
A) 0
B) 1
C) 2
D) 3
E) 5
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these electron energy level patterns would absorb light with the shortest wavelength 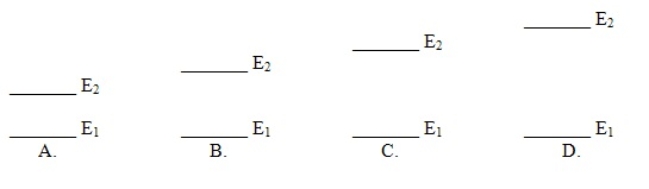
A) A
B) B
C) C
D) D
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 92
Related Exams