A) [Ne]3s2
B) [Ne]3s23p6
C) [Ar]4s13d1
D) [Ar]4s2
E) [Ar]3d2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Not Answered
Which element has the following ground-state electron configuration? 1s2 2s2 2p6 3s2 A)Na B)Mg C)Al D)Si E)Ne
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the element with the electron configuration [Xe]6s24f7.This element is
A) a halogen.
B) a lanthanide element.
C) a nonmetal.
D) an actinide element.
E) a noble gas.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these pairs consists of isoelectronic species?
A) Mn2+ and Ar
B) Zn2+ and Cu2+
C) Na+ and K+
D) Cl− and S
E) K+ and Cl−
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An isoelectronic series is
A) a series that has two or more species that have identical nuclear charges, but have different electron configurations.
B) a series that has the same ionization potentials.
C) a series that can have only up to three species and have similar electron configuration and similar nuclear charges.
D) a series that has two or more species that have identical electron configurations, but different nuclear charges.
E) a series that has the same nuclear charge.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are in the 4p orbitals of vanadium?
A) 0
B) 2
C) 4
D) 5
E) 6
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct electron configuration for sulfur (Z = 16) .
A) 1s21p62s22p6
B) 1s22s22p83s23p4
C) 1s22s22p83s23p2
D) 1s22s22p63s23p4
E) 1s22s22p63s23d4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the smallest atomic size?
A) Na
B) Ar
C) K
D) Ca
E) Kr
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Not Answered
An element with the general electron configuration for its outermost electrons of ns2np1 would be in which element group? A)2A B)3A C)4A D)5A E)8A
Correct Answer

verified
Correct Answer
verified
Short Answer
The orbital diagram for a ground-state carbon atom is 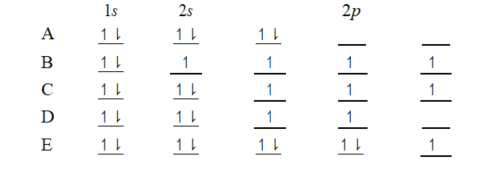
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ground-state atom has an electron configuration described by the following orbital diagram? 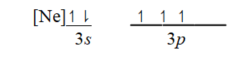
A) Phosphorus
B) nitrogen
C) arsenic
D) vanadium
E) sulfur
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ground-state electron configuration for an atom of indium is
A) [Kr]5s24p64d5
B) [Ar]4s23d104p1
C) [Ar]4s24p63d5
D) [Kr]5s25p64d5
E) [Kr]5s24d105p1
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis dot symbol consists of the symbol for the element surrounded by dot(s) .What does the symbol represent?
A) Electron configuration
B) Valence electrons
C) Atomic number
D) Atomic mass
E) Nucleus and core electrons
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electronic structure 1s22s22p63s23p64s23d8 refers to the ground state of
A) Kr.
B) Ni.
C) Fe.
D) Pd.
E) None of these choices is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these atoms has the smallest radius?
A) Al
B) P
C) As
D) Te
E) Na
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the emission of light at only specific wavelengths?
A) Emission spectra
B) Hydrogen spectrum
C) Wave spectra
D) Limited spectra
E) Line spectra
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The size of an atomic orbital is associated with
A) the principal quantum number (n) .
B) the angular momentum quantum number (l) .
C) the magnetic quantum number (ml) .
D) the spin quantum number (ms) .
E) the angular momentum and magnetic quantum numbers, together.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the elements listed below has the greatest atomic radius?
A) B
B) Al
C) S
D) P
E) Si
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ion is isoelectronic with Ar?
A) Fe2+
B) F−
C) Br−
D) Ga3+
E) Ca2+
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
The orbital diagram for a ground-state oxygen atom is 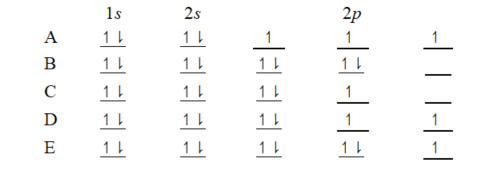
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 107
Related Exams