A) There is no movement of electrons from one form to another.
B) Resonance structures are not isomers.
C) Resonance structures differ only in the arrangement of electrons.
D) Resonance structures are in equilibrium with each other.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules are constitutional isomers?
A) I, II, IV
B) II, III, IV
C) I, III, IV
D) I, II, III
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds are non-polar?
A) I, IV
B) I, II
C) II, III
D) II, IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Convert the following skeletal structure to a condensed structure. 
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the electron geometry around the indicated atom in each species. 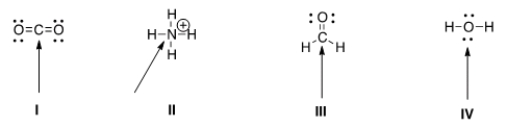
A) I = Linear; II = tetrahedral; III = trigonal planar; IV = tetrahedral
B) I = Linear; II = tetrahedral; III = trigonal planar; IV = linear
C) I = Trigonal planar; II = linear; III = tetrahedral; IV = trigonal planar
D) I = Tetrahedral; II = trigonal planar; III = linear; IV = tetrahedral
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formal charge of carbon in carbon monoxide (CO) when drawn with a triple bond?
A) 0
B) -2
C) -1
D) +1
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecule has the greatest difference in electronegativity (E) between the two different elements?
A) CO2
B) H2S
C) NH3
D) H2O
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following Lewis structures is correct? 
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a resonance structure of the compound below? 
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atomic orbitals overlap to form the C-H bonding molecular orbitals of acetylene, C2H2?
A) Csp + H1s
B) C2p +H1s
C) Csp3 + H1s
D) Csp2 + H1s
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the appropriate conversion of (CH3) 2CHCH2CHClCH3 to a skeletal structure? 
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization of the nitrogen atom in the ammonium cation, NH4+?
A) sp3
B) sp2
C) sp
D) p
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many constitutional isomers are there for a molecule having the molecular formula C2H4Cl2?
A) 1
B) 2
C) 3
D) 4
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following in order of decreasing importance as contributing structures to the resonance hybrid of formaldehyde, H2CO. 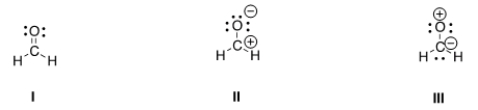
A) I > II > III
B) I > III > II
C) II > I > III
D) III > II > I
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules contain both covalent and ionic bonds?
A) I, II
B) I, IV
C) II, III
D) II, IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ground-state electronic configuration of a fluorine atom?
A) 1s2, 2s2, 2p2
B) 1s2, 2s2, 2p3
C) 1s2, 2s2, 2p4
D) 1s2, 2s2, 2p5
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has polar covalent bonds?
A) MgO
B) NH3
C) Cl2
D) NaBr
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ground-state electronic configuration of a carbon atom?
A) 1s2, 2s2, 2p5
B) 1s2, 2s2, 2p2
C) 1s2, 2s2, 2p6
D) 1s2, 2s2, 2p4
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which compound contains the most polar bond? I II III IV
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following Lewis structures is correct? 
A) I, II
B) I, III
C) II, III
D) III, IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 70
Related Exams