A) 2
B) 4
C) 6
D) 7
E) 8
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many dots does the Lewis dot symbol for sodium have around it?
A) 1
B) 2
C) 0
D) 3
E) 7
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
A triple bond arises when two atoms share ___________ pairs of electrons.
Correct Answer

verified
Correct Answer
verified
Essay
Describe,with appropriate explanations,the key factors which affect the magnitude of the lattice energy of an ionic substance.
Correct Answer

verified
By Coulomb's law,the energy of two elect...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Identify the correct equation to calculate the formal charge on an atom.
A) formal charge = valence electrons - lone pair electrons
B) formal charge = valence electrons - associated electrons
C) formal charge = valence electrons - bonding electrons
D) formal charge = bonding electrons + lone pair electrons
E) formal charge = valence electrons - bonding electrons + lone pair electrons
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
All of the following molecules contain only single bonds except
A) H2O2.
B) Cl2.
C) CH4.
D) H2O.
E) SO2.
G) A) and E)
Correct Answer

verified
E
Correct Answer
verified
True/False
A double bond cannot exist between a carbon atom and an oxygen atom.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the Lewis structure of the iodate ion,IO3-,that satisfies the octet rule,the formal charge on the central iodine atom is
A) 2.
B) 1.
C) 0.
D) -1.
E) -2.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many dots does the Lewis dot symbol for chlorine have around it?
A) 1
B) 2
C) 5
D) 7
E) 17
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formal charge on phosphorus in a Lewis structure for the phosphate ion that satisfies the octet rule?
A) -2
B) -1
C) 0
D) +1
E) +2
G) A) and E)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which of the following represents the most polar bond?
A) B-C
B) S-O
C) C-O
D) B-O
E) C-C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following species does the central atom violate the octet rule?
A) CH4
B) SF4
C) PCl4+
D) CCl3+
E) NH3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Describe in brief how electronegativity values can be used to predict the percent ionic character of a bond between two atoms.
Correct Answer

verified
Take the electronegativity difference (Δ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
In which one of the following structures does the central atom have a formal charge of +2?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of resonance structures for the nitrate ion that satisfy the octet rule is
A) 1.
B) 2.
C) 3.
D) 4.
E) none of these
G) B) and D)
Correct Answer

verified
C
Correct Answer
verified
True/False
Ionic compounds may carry a net positive or net negative charge.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formal charge on the fluorine in ClF2+?
A) -1
B) +1
C) 0
D) -2
E) -3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following Lewis structure for ClO3F,chlorine has a formal charge of ____ and an oxidation number of ____. 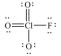
A) 7,7
B) 7,-1
C) 1,1
D) 1,-1
E) 1,7
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis dot symbol consists of the symbol for the element surrounded by dot(s) .What does the dot or dots represent?
A) Electron configuration
B) Valence electrons
C) Atomic number
D) Atomic mass
E) Core electrons
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 95
Related Exams